Regeron Pharmaceuticals is Changing Drug Discovery
How an entrepreneur, a research genius and an industry legend built this century's most inventive new drug company. Together they just might change medicine


There is a 40-something aerobics instructor in a Dallas suburb whose cholesterol level is so low—a sixth that of a normal person—that scientists think she is nearly certain to be spared heart disease, no matter what she eats. In Berlin, Germany, a small child arrived at the hospital with amazingly well-developed muscles. At the age of four, he could hold a seven-pound dumbbell in each of his outstretched hands.
These people—genetic mutants seemingly out of an X-Men story—are more than just blessed by nature. They are leads to the future of human health, as pursued by one of the most prolific drug hunters of his generation: George Yancopoulos, 53, the chief scientific officer of Regeneron Pharmaceuticals. An experimental treatment he created based on the heart mutation is among the hottest in the industry. A second based on the buff toddler might help cancer patients whose muscles are wasting.
Other Yancopoulos medicines—for asthma, rheumatoid arthritis and cancer—are in testing, too. All are based on a unique method his team has developed of putting large swaths of human DNA in mice and using the rodents to quickly generate human drugs.
“This is why you get into this business,” says Yancopoulos. “You think you understand biology. You think you have an insight. And you think it might cure disease.”
Three of his medicines are already on the market, an amazing feat in an industry where researchers toil a lifetime to develop a single drug and get treated as superstars if they manage two. Alas, of the three, two were commercial duds: A treatment for a rare genetic disorder, and a cancer drug similar to Genentech’s Avastin. But his big hit came in late 2011 with a medicine called Eylea that treats the leading cause of adult blindness. At a time when Wall Street was convinced that the pharmaceutical industry had run out of blockbuster products, it generated $838 million in its first full year, and sales are expected to jump 55 percent this year to $1.3 billion.
But it isn’t just smart science that’s turned Regeneron into one of the world’s most innovative companies (it ranks No. 4 on Forbes’s annual list this year). It’s also smart business. One key reason Yancopoulos has been able to succeed has been the support of his boss, founder and Chief Executive Leonard Schleifer, 61, an MD-PhD who has turned out to be one of biotech’s shrewdest dealmakers and who, for two decades, has protected Yancopoulos from investor demands for results as failures piled up. “George was too talented,” says Schleifer. “The people around him were too talented. It wasn’t a matter of whether we can do this. It was a matter of when we can do this. Would we survive long enough to have a hit?”
They have, and then some. Against the grain of industry trends, the company has found ways to create effective new treatments on bargain budgets. According to a Forbes analysis of 220 drugs approved over the past decade for publicly traded companies, the companies that invented three or more medicines spent an average $4.3 billion in R&D per drug. The big boys spend still more: $5.5 billion for Merck, $7.8 billion for Pfizer and $10 billion for Sanofi, Regeneron’s partner in many of its projects. Regeneron’s cost per drug? Only $736 million. “He’s been successful beyond anything I could imagine,” says Fred Alt, the Harvard Medical School geneticist who first told Schleifer about Yancopoulos.
Accordingly, Schleifer is approaching billionaire status. He’s worth $800 million, largely in Regeneron stock, according to Forbes estimates. Meanwhile, Yancopoulos has made more money than almost any research biologist in history. Last year, he received a $82 million pay package, also mostly stock, which bettered every chief executive in America except Oracle’s Larry Ellison. His estimated total net worth: $400 million.
But for all his success, medicinally and financially, Yancopoulos has yet to create a drug that has really changed the world. His new projects—the heart medicine, the asthma drug—might just be the kind that save thousands of lives or become household names.
“Everything that we’ve been doing for 25 years, it interconnects,” says Yancopoulos. “It’s not like we changed direction in the middle, or we did a new trick. It’s all building on the foundation of those early ideas, and we’re just taking them to the next level.”
Regeneron’s roots lie in a Chinese restaurant. On the Upper East Side of Manhattan, in 1988, Schleifer, an assistant professor of neurology at Weill Cornell Medical College, scrawled a deal on a napkin with a Merrill Lynch venture capitalist and walked out with $1 million in funding, a chief executive job and a new company aimed at healing nerves. He then recruited top scientists to serve as advisors, including three Nobel Prize winners who joined Regeneron’s board of directors.
One of the advisors told Schleifer that Yancopoulos, then a 28-year-old professor at Columbia, was “the young superstar of his generation”. Yancopoulos also had a unique reason to listen to Schleifer’s overtures. His dad, a Greek immigrant who had come to America hoping to re- build the fortune his father (kept in slavery in Turkey before escaping to build some of Greece’s first electric power plants) had lost to the Nazis. He hated that his brilliant son had chosen a career that paid as poorly as academia. Yancopoulos had $2 million in grants that would fund him for eight years, but only $35,000 made it into his pocket. America, his father told him, should pay him a lot more.
Yancopoulos went as far as scouting potential laboratory locations with Schleifer but wouldn’t actually sign on the dotted line. Finally, at a meeting at an Italian restaurant in New York’s Westchester County, Yancopoulos showed up with his dad. “His father wanted to interview me, rather than me convincing George,” says Schleifer. Dad approved, and Yancopoulos signed up.
But the eight scientists who had agreed to work with Yancopoulos at Columbia weren’t willing to follow. In those days, going to a company meant throwing away an academic career. They weren’t alone. It took months for Yancopoulos to make his first hire, a jobless theoretical physicist interested in biology, and two years to build out his core research squad. Hiring slowly and correctly, however, paid off.
That original group of a half-dozen has partnered with Yancopoulos on every breakthrough, and almost every one of them remains at Regeneron to this day. “The techniques being applied were really cutting-edge,” says Neil Stahl, Yancopoulos’ right-hand man since 1991. “I became convinced that I could do more science here than as a lonely assistant professor someplace.” Initially, the path to glory seemed simple. Yancopoulos figured out that the growth-factor proteins, key chemical signals that spur healing, were much the same in the brain as elsewhere in the body. Isolate one, put it in the brains of patients and—wham!—they’d help patients with Lou Gehrig’s disease. Schleifer imagined the two of them, triumphant, getting permanent seats in Yankee Stadium by first base, where Gehrig played, and counting their money as patients’ lives were transformed. Instead, the drug failed in clinical trials.
Dejected, Schleifer made an off-the-wall suggestion to Yancopoulos: “Why don’t we just call up Roy Vagelos? Maybe he’ll help us out.”
It was an audacious idea. Then chief executive of Merck, Vagelos was, thanks to drugs that had changed the care of heart patients and cured river blindness in Africa, one of the most well-known and respected executives in the history of the pharmaceutical industry—and Yancopoulos’ hero. The timing was fortuitous. Vagelos was on his way out at Merck amid a management shake-up and agreed to meet with the team, if they could do it immediately. Yancopoulos pulled an all-nighter, throwing together a presentation. It worked. Vagelos saw potential. He became Regeneron’s chairman. “They have a terrific CEO and a terrific head of research in that company,” says Vagelos. “And you know, that’s the answer in our industry. The CEOs are important, but without new products CEOs are in trouble.”
And so the Vagelos school began. Lesson one: Stop betting on drugs when you won’t have any clues they work until you finish clinical trials. (That ruled out expanding into neuroscience—and is one of the main reasons other companies are abandoning ailments like Alzheimer’s.) Lesson two: Stop focusing only on the early stages of drug discovery and ignoring the later stages of human testing. It’s not enough to get it perfect in a petri dish. Regeneron became focussed on mitigating the two reasons that drugs fail: Either the biology of the targeted disease is not understood or the drug does something that isn’t expected and causes side effects.
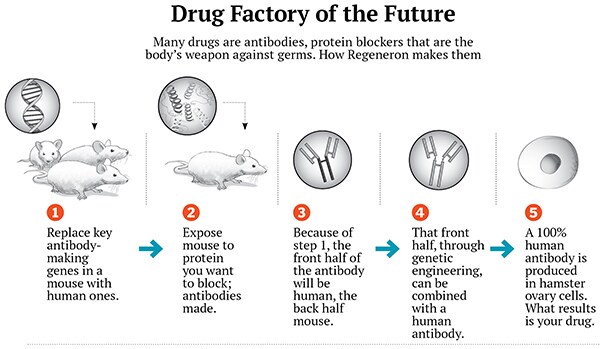
There were still more failures: A second Lou Gehrig’s drug, a weight-loss drug. Then a new technology emerged out of their efforts to discover growth factors, which work by tripping switches, called receptors, on the outside of cells. At the time, it was easy to find receptors but hard to find growth factors. Yancopoulos’s first hire, the physicist, discovered a way to fish out the right growth factor using the receptor. And then Yancopoulos figured out how to make these receptor-trawlers into a trap that would catch all the growth factor in the blood, blocking it. Yancopoulos called the drugs “Traps”.
Regeneron’s first Trap, for a rare kidney disease, hit the market in 2008. And while it generates just $20 million in annual sales, it paved the way for Zaltrap, for cancer, which generated $14 million in the first quarter, and Eylea, the fast-selling blockbuster for age-related macular degeneration. Eylea’s success has sent Regeneron shares up more than 400 percent since its November 2011 approval. “I was thinking they’d come in higher than expectations but not that much higher,” says Biren Amin, an analyst at Jefferies. “It was phenomenal.”
But fishing out growth factors and laboriously creating Trap drugs was only the first step. Uniquely, and distinct from his mentor, Yancopoulos thinks creating technologies that make drug discovery easier is the key to inventing new medicines. And to really make sure that his medicines have a good shot at succeeding, Yancopoulos needed to invent a better way of understanding how the body worked and of making targeted medicines to affect it. Not just a better mousetrap but better mice.
The problem with lab mice is simple: Despite a shocking amount of genetic similarity, their tiny bodies don’t always work like those of humans. It’s a cliché that drugs that shrink tumours in mice don’t do the same in people. He and his team developed a method of putting up to 6 million letters of human DNA at a time into a mouse genome. The National Institutes of Health is paying Regeneron to use this technology to discern the function of 3,500 genes.
The mice opened up an even bigger opportunity. Many of the biggest drugs of the biotech age, including Herceptin for breast cancer, Rituxan for lymphoma and Humira for rheumatoid arthritis, are antibodies, smart bombs the immune system uses to neutralise germs. These drugs are traditionally made in mice and then laboriously converted, amino acid by amino acid, into antibodies the human body will accept. But with Yancopoulos’ mice, it was possible to replace part of the mouse immune system with human genes. This meant that antibodies could be quickly made in mice and, with a simple step, turned into drugs ready for humans.
Sanofi, seeing the potential of this technology, in 2007 agreed to pay Regeneron $100 million a year in R&D funding for rights to 50 percent of profits from the antibody drugs that resulted. Five drugs entered clinical trials in the first two years, at which point Sanofi upped funding to $160 million a year, as well as agreeing to handle much of the clinical testing and sales and marketing. Sanofi shares in the rights to the cholesterol drug, asthma drug and Regeneron’s other experimental prospects.
For Regeneron (and Sanofi) to really benefit from this new system, they need more than just mouse genetics: They need mutants. The best proof that a drug target works in people comes from combining the mouse data with human genetics.
How might this actually work? Look no further than a gene called PCSK9, which first turned up in patients in France who had superhigh cholesterol, putting them at risk for heart attacks at a young age. Usually, mutations that break a gene are more common than those that make it work better. So researchers at the UT Southwestern Medical Center searched a giant database of heart patients for people with low cholesterol. They learned that one group that had one broken copy of the PCSK9 gene had 28 percent lower cholesterol levels and an 88 percent reduction in their risk of heart disease. There are several people walking around with two broken PCSK9 genes—including the aerobics instructor with super-low cholesterol. All are healthy but have low-density-lipoprotein levels of 15 mg per deciliter, compared with 100 for most healthy people.
It’s basic math: Turning a gene up is bad, and knocking it out is good, and if really knocking it out is really good, then a drug to block it should be good, too.
Regeneron and Sanofi beat Amgen into clinical trials to prove the thesis. Will patients readily take an injection to control high cholesterol? “It’s the debate of the decade in the pharma industry,” says cardiologist Steven Nissen of the Cleveland Clinic, who is working with Amgen. He thinks that the PCSK9 drugs will do well but will be mega-hits only if they prove they can reduce heart attacks and strokes beyond what can be done with statins, which are now cheap generics. If there is a flaw, it is, as with many of Yancopoulos’ drugs, that he went into an area with too many competitors: Pfizer, Alnylam and Roche are all working on PCSK9, too.
If he succeeds at getting a few more hits, Yancopoulos has a chance at joining a pantheon of larger-than-life drug researchers who, in the 1960s and 1970s, invented whole categories of medicine—like Leo Sternbach at Roche, who invented Valium and six similar drugs, and Merck’s Maurice Hilleman, who developed more than 36 vaccines—and silencing comments from colleagues who wonder what he’s been building all this time. “George Yancopoulos is an awesome scientist,” says biologist and venture capitalist Corey Goodman. “Whether that first-rate molecular biology translated into drugs is another matter.”
Yancopoulos’ drug based on the muscular toddler, in early stages of development, seems a no-brainer he thinks other similar antibodies failed because they weren’t good enough. There’s also a rheumatoid arthritis drug. But he’s most excited about a medicine to treat allergies and asthma, problems he says barely existed when he was a kid. People with certain mutations in the gene it targets get sicker, and early studies in humans show promise. Results for more than a dozen drugs could roll in by the end of the decade, a long grind in many industries but a sprint in terms of drug discovery.
Yancopoulos is looking even further out. The ability to cheaply decode human DNA will launch a whole new set of experiments for his mice and targets for his antibodies. “We’ve been doing genetics here for 25 years from day one it’s just the next step,” he says. “It’s the more powerful way to do it.” And as with stepping up from Traps to antibodies, he says that now his drug development engine is on “the precipice of change.” The next major leap in medicine? It’s only a matter of time.
Entrepreneurs Clinic
“We are just not that smart”Regeneron CEO Leonard Schleifer on what every startup can learn from a science-based business  THE MAJORITY OF YOUR BOARD OF DIRECTORS ARE TOP SCIENTISTS. WHY IS THAT IMPORTANT?
THE MAJORITY OF YOUR BOARD OF DIRECTORS ARE TOP SCIENTISTS. WHY IS THAT IMPORTANT?
I believe that companies rot, and they rot from the top down. Too often the keys to the kingdom are given to commercial folks who don’t value long-term research. When you don’t value something, you don’t get good results from it, and the bottom line is that then, all of a sudden, the long term becomes the short term, and you don’t have anything.
HOW DO YOU KEEP A PARTNERSHIP GOING FOR 25 YEARS?
One of the mistakes that people make is they think that they can be not only the chief scientist but also the chief operating, the chief scientific, chief medical officer and the chief inventor. It’s a big ego thing that young people think they can do it all. George clearly had the science genius, and I had the flair, I think, for a lot of the entrepreneurial sort of stuff. But he’s also an entrepreneur, and I can be a scientist.
INVESTORS OFTEN WANT COMPANIES TO BE FOCUSSED ON ONE THING: A SINGLE, MOST IMPORTANT DRUG OR A NEW TECHNOLOGY. YOU’VE REFUSED TO DO THAT.
“Focus” is a dirty word for us, okay? It’s a big mistake to think that you can pick the very best thing that you should focus on and then ignore all the other things. Wouldn’t it be wonderful if we could pick only the things that work in our business? Amgen’s new CEO, I heard, said they only were going to work on the things that work. Good luck to him. We are just not that smart.
YOU’RE A CONSUMMATE DEALMAKER, EVEN ONCE CONVINCING SANOFI TO PAY YOU WHEN THEY GAVE THE RIGHTS TO A DRUG BACK TO YOU. WHAT’S YOUR SECRET?
When we negotiate, we actually care what happens five or ten years down the road. A lot of people make a deal, and they want to know what happens to my stock this week, maybe next month, and they are willing to mortgage their future for short-term success. We never did that. We were unwilling just to give it away because we knew at the end of the day these companies we were working with, they needed these things that we had.
First Published: Sep 17, 2013, 07:23
Subscribe Now