India’s in the middle of a second wave of the Covid-19 outbreak. Over the past week, active cases in the country have been on the upswing, and daily cases have been crossing 50,000 regularly. The last time that happened was in October 2020, when India was emerging out of its first wave. Since then, cases had dropped significantly, even falling below 10,000 daily.
Over the last week, however, things have taken a turn for the worse. “After having successfully brought down the number of new Covid-19 cases from mid-September to February, India is now witnessing a rapid rise in cases,” health secretary Rajesh Bhushan said in a letter to the chief secretaries of all the state governments on March 30. “The current rise in cases is of concern and has the potential of overwhelming health care systems, unless checked right now.”
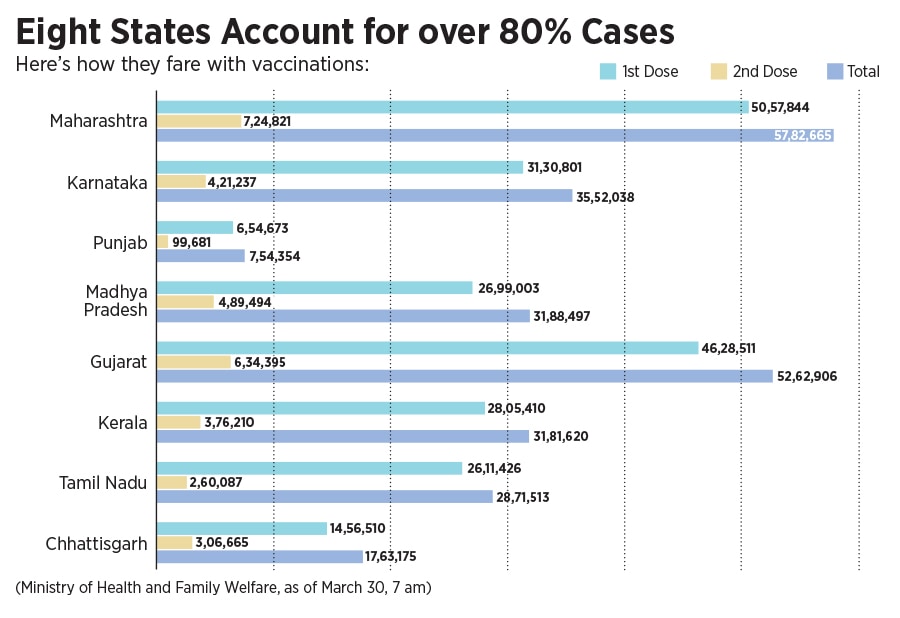
On March 28, financial capital Mumbai announced a strict night curfew, and the state government is even contemplating a lockdown in Maharashtra. Several other states have warned of and taken stringent measures, including curfews, to curtail the spread of cases. Currently, eight states account for about 80 percent of the cases, a number that is only expected to swell in the coming days. The central government has also identified urban clusters including Delhi, Mumbai and Bengaluru, as a cause of concern in the weeks ahead.
The worries have been corroborated by India’s largest bank, the State Bank of India (SBI), which has said in a report that India is witnessing a second wave beginning February 2021. “Considering the number of days from the current level of daily new cases to the peak level during the first wave, India might reach the peak in the second half of April,” says SBI in the report published on March 25. “The entire duration of the 2nd wave might last up to 100 days counted from 15th February.” That could very well mean the crisis isn’t likely to end before May.
The second wave comes at a time when India has decided to make all its citizens aged above 45 eligible for vaccinations. So far, since the programme began on January 16, vaccines were only provided to those on the frontlines before it was extended to those above 60 years. On March 1, those above the age of 45, but with co-morbidities, were also allowed to vaccinate themselves. India has approved the use of Covishield, commonly known as the AstraZeneca vaccine, and the indigenous Covaxin, developed by Hyderabad-based Bharat Biotech, currently to vaccinate its people.
![vaccination-1 vaccination-1]()
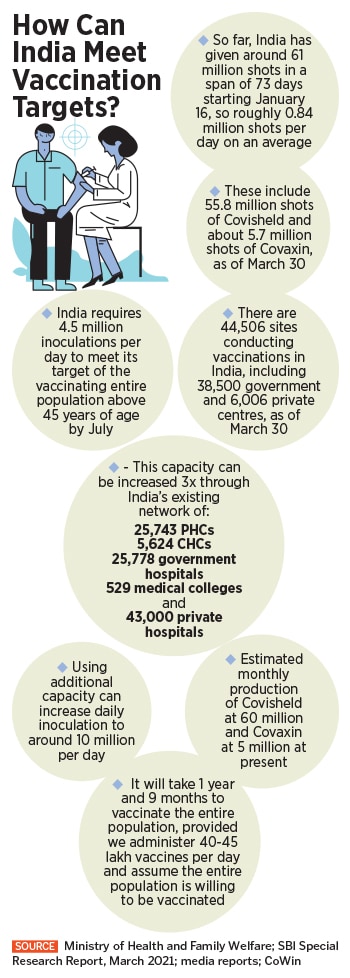
So far, some 61 million doses of vaccines have been administered, according to the government, of which 8.4 million people have completed both the first and second dose. That’s only about 0.6 percent of the Indian population of 1.35 billion people. Additionally, some 53 million people have received the first dose so far. That number is in stark contrast to what the Indian government had initially set out to achieve by July when India began the vaccination programme. The country is looking to vaccinate between 200 million and 250 million people by July 2021, four months from now.
“We can’t force anyone to get vaccinated,” says RS Sharma, the CEO of the National Health Authority, the agency responsible for the vaccination registration. “There are no problems when it comes to supply or constraints in logistics. Demand has been rising, and while we were doing 3 million vaccinations a day, we are looking to raise that to five million a day. The system can handle up to 20 million registrations a day.”
Laggard vaccination
![vaccination-2 vaccination-2]()
Yet, India is still vaccinating at less than 1.5 million doses a day across its 44,500 centres. A total of 63 million registrations have been received so far for vaccinations. On March 30, 1.29 million doses were administered, down from the high of 3 million on March 22.
“If we assume more number of people are willing to take vaccines and the daily inoculation increases to 4-4.5 million from the current maximum level of 3.4 million, then with this capacity we can vaccinate our population above 45 years in 4 months from now,” the SBI report goes on to add.
So far, the government has ordered some 141 million doses of vaccine from the Serum Institute of India, since the vaccination began. With Bharat Biotech, the government had initially placed an order of 5.5 million doses. As per a PTI report earlier in March, the company has received an order for an additional 4.5 million doses, out of which over eight lakh will be supplied to countries like Mauritius, Philippines and Myanmar free of cost as a goodwill gesture. Bharat Biotech has also donated 1.65 million doses to the government, as per the report.
The regulator Drugs Controller General of India (DCGI) is yet to approve the Sputnik V vaccine, even though the vaccine has been administered in around 30 countries, including Russia, Belarus, Argentina, Bolivia, and the UAE so far.
Approval for Sputnik can certainly add more arsenal to the national capacity. Apart from Dr Reddy’s, Hyderabad-based Virchow Biotech Private Limited will also produce 200 million doses of Sputnik V, while Bengaluru-based Stelis Biopharma has also decided to produce 200 million doses. Besides, Hyderabad-based Gland Pharma will produce 252 million doses and Hyderabad-based Hetero Drug will produce 100 million doses in the coming months.
A subject expert committee (SEC) of the health ministry is likely to meet on March 31 to consider granting emergency use authorisation to the Sputnik V manufactured by Dr Reddy after the company had submitted the immunogenicity data. The SEC is part of the Central Drugs Standard Control Organisation (CDSCO) headed by the Drugs Controller General of India.
In addition, a vaccine manufactured by the Serum Institute of India, in partnership with UK based Novavax, Covavax is undergoing trials in India, even though the vaccine has been found to provide 89 percent efficacy in its Phase III trials. The company now expects the vaccine to be rolled out from September.
A fifth vaccine, manufactured by Ahmedabad based Zydus Cadila, will only be considered for approval once the company submits the data of its Phase III trials. The company had earlier expected to complete the process by March end and laid out plans to manufacture some 200 million doses. For now, the company is still waiting for all its dosings to be completed. Zydus’s candidate, called ZyCov-D, will be the second indigenous vaccine after Bharat Biotech’s Covaxin.
“There will be more and more vaccines passing the regulatory requirements for emergency usage and adding to the vaccine supply available,” Satyajit Rath, a scientist at the National Institute of Immunology and the adjunct professor at the Indian Institute of Science Education and Research had told Forbes India earlier. “The logistical catch is going to be how to decide which vaccine is to be supplied where and when, for the most optimal vaccine distribution plan.”
![vaccination-3 vaccination-3]()
A November 2020 report from Credit Suisse says that India will need about 1.7 billion Covid-19 vaccine doses to inoculate the majority of its adult population. “India targets to administer 400-500 million doses by July-2021,” the report says. “In our view, there is sufficient capacity for vaccine manufacturing (>2.4 bn doses) and various components like vials, stoppers, syringes, gauze, alcohol swabs, etc.”
Yet, the government has not allowed largescale private sector participation in the programme. “About 70 percent of the health care capacity in India is held by the private sector and they can be instrumental in reducing hesitancy and speed up the process,” Viren Shetty, the COO of Narayana Health had told Forbes India. “It would be far more helpful to allow the private sector to procure vaccines and offer options. There are enough manufacturers to procure vaccines from.” Alongside, India had also embarked on an ambitious global vaccine export programme to send vaccines to countries across the world. According to the ministry of external affairs, India has exported more than 60 million doses since January 20.
“We should not simplify vaccination as merely a supply chain issue. It"s a much more complex task that needs community engagement, private sector engagement and involvement of NGOs and activists,” says Prashanth N Srinivas, assistant director (research) at the Institute of Public Health, Bengaluru. “For example, there are village health, sanitation and nutrition committees under the National Health Mission. How many of them have met and discussed how to ensure vaccination coverage of our villages? How do you involve all of them in your engagement efforts?”
Long Way to Go
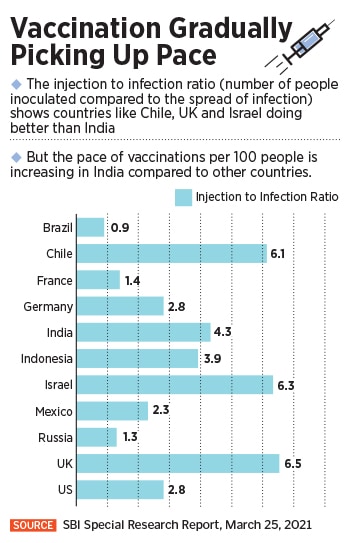
Now, even as the second wave surges, expectations are that it may not be as bad as the first one. “Though global Covid-19 experience shows a second wave is much higher in intensity than the first wave, the presence of vaccine makes the difference currently,” the report by SBI says. “Thus India will be able to manage the situation better.”
Yet, India continues to face concerns of vaccine hesitancy among its population. “One of the main problems in our approach has been that people are just passive recipients of our awareness and education activities,” Srinivas says. “There is vaccine hesitancy even among those who are more educated. On the other hand, we have had a huge access issue in our health system, even before Covid.”
That means, there are, clearly, too many factors that have to fall in place for the vaccination targets to be achieved: Willingness of people to get vaccinated, the supply of vaccines and capacity of the public and private healthcare systems to accommodate more people to vaccinate per day. Samiran Panda, head, epidemiology and communicable diseases at the Indian Council of Medical Research (ICMR), reckons that while vaccine hesitancy slowed down the first phase of vaccination, more people across socio-cultural backgrounds and age groups are becoming more open to getting vaccinated given the emerging threats of new variants of the coronavirus and the second wave.
“Apart from government efforts, community engagement and their awareness are important for vaccination to be successful. We have to help people understand the value of getting vaccinated away from confusing messages in the social media and the lack of awareness,” says Panda, who is also a member of the government’s National Expert Group on Vaccine Administration for Covid-19 (NEGVAC). According to him, for the pace of vaccinations to increase, there needs to be multiple vaccines available in the market “apart from just Covisheid and Covaxin”. Panda, however, did not comment on the approval process and indicative timelines for the other vaccine candidates
Meanwhile, the discovery of newer variants of the virus in India is also likely to push up vaccinations in the country. The health ministry had last week said that a variant with two mutations—E484Q and L452R—that make them better at evading the body’s defences was found in the country. In addition, the UK, Brazilian and South African variants have also been circulating here.
Dr Pavan Kumar Gorukanti, director of Yashoda Hospitals in Hyderabad that has vaccinated 29,835 people as of March 27, agrees with Panda that due to the possibility of a second wave, vaccination compliance is already increasing. “People are now realising that the benefits far outweigh the risk. The demand for it has exceeded the supply, which is a positive sign,” he says. “For the small majority of people getting infected with Covid after the vaccinations, the symptoms observed were very mild with almost no one reported serious cases requiring ICU care or similar.”
![vaccination-4 vaccination-4]()
That has also meant that people have been shying away from taking the second dose after receiving their first one. For instance, out of 5-odd crore people who have received the first dose in India, only 89 lakh have received the second dose. In Telangana, where Dr Gorukanti’s hospital is located, the state health ministry on March 18 said that against 3 crore people who got their first dose, only 65 lakh people turned up for their second one. Dr Gorukanti believes this trend is reversing. “Initially, compliance for the second dose was low, likely due to the low and falling incidence of Covid cases in India at that time. However, Covid cases are rising again, and compliance for the second dose is significantly increasing,” he says.
The other two key aspects of the vaccination game is supply and capacity to ramp up daily numbers. At present, as per the Co-Win website on March 30, 44,506 sites, including 38,500 government and 6,006 private centres, are vaccinating across the country. “With more vaccinations getting approved, the government should sign contracts in advance of clinical
approval, thus shouldering some of this risk and incentivising firms to install capacity earlier,” adds the SBI report.
“The responsibility has been given to authorities and officials, but you can"t robotise vaccination and expect people to queue up,” adds Srinivas. “We have to build outreach into communities and people that are embedded into the remotest of areas. These are things that have to be planned not even at the state level, but by involving local panchayats. Gram panchayat members need to be able to decide where vaccination points should be. Imagine the health ministry is taking that call... what works for Nagaland, will it work for Tamil Nadu or Goa?”
India, as per the SBI report, has over 1,00,674 potential centres, three times of what is being used for vaccinations. These include 25,743 primary health care centres (PHC), 5,624 community health care centres (CHC), 25,778 government hospitals, 529 medical colleges and close to 43,000 private hospitals. “Going by the health infrastructure, we have the capacity to increase the daily doses of around 1 crore per day from the current 34 lakh per day,” the report states.
“We will not be able to achieve targets in a very predictable manner,” adds Srinivas. “We have to do much more than just involve officials. We have to involve the community, the private sector, NGOs, and really decentralise immunisation.”
A second wave is here and only a collective effort can salvage the situation.