The world’s largest Covid-19 vaccination programme will begin on Saturday, January 16.
At 10:30 AM on January 16, Prime Minister Narendra Modi will officially launch the first phase of Covid-19 vaccination, involving a vaccine developed by AstraZeneca and the indigenously developed vaccine from Bharat Biotech. The phased rollouts will initially vaccinate ten million health care and 20 million frontline workers, followed by 270 million senior citizens and people with co-morbidities like diabetes, hypertension, organ transplants etc. In all, 300 million people are going to be inoculated by July this year.
That’s roughly a little more than 20 percent of the country’s 1.3 billion population. “Prime Minister Narendra Modi will launch the pan-India rollout of Covid-19 vaccination drive on 16th January, 2021 at 10:30 AM via video conferencing,” a statement from the Indian prime minister’s office said.
The vaccination programme will be held over 3,000 session sites across the country, and around 100 beneficiaries will be vaccinated at each session site on the inaugural day. India is currently the world’s second worst-hit Covid-19 country with over 10.52 million cases. Of this, over 213,000 cases are currently active.
“This will be the world’s largest vaccination programme covering the entire length and breadth of the country,” the prime minister’s office said.
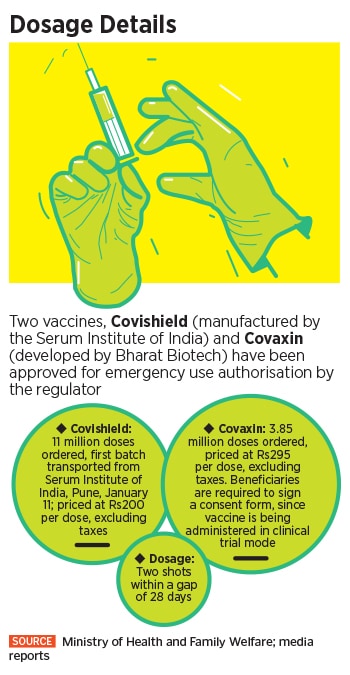
To undertake the exercise, the government has already purchased 11 million doses of Covishield, manufactured by the Serum Institute of India, at around Rs 200 per dose, excluding tax. In addition, the government has also paid Rs 295 per dose excluding tax for 3.85 million doses of Covaxin, manufactured by Hyderabad-based Bharat Biotech. The company has also offered to supply 1.65 million doses for free as a “special gesture” to the government.
The Serum Institute of India currently has a stockpile of about 50 million doses—with plans to manufacture 100 million doses monthly in 2021—while Bharat Biotech has plans of manufacturing 700 million doses in 2021 at its four facilities across the country. The company has a stockpile of about 20 million doses.
“This is the initial lot of supply of vaccine doses and would be continuously replenished in the weeks to come,” the health ministry said on January 14. “Therefore, any apprehension being expressed on account of deficient supply is totally baseless and unfounded.” For now, the vaccination is allowed only for those who are above the age of 18 years, while women who are pregnant or unsure about their pregnancy will not receive the vaccine. Interchangeability of the vaccine is also not permitted--the second dose of the vaccine should also be of the one that was administered as first dose.
The expenses towards the vaccines, meanwhile, will be borne by the Central government for now. “It has been decided that state governments will not have to bear the expenses of vaccination of these 3 crore (30 million) people in the first phase. The Government of India will bear these expenses,” Modi had said on January 11.
All Eyes on the States
The last leg of the vaccine distribution to session sites will be undertaken by the Centre and the state governments, with ministries other than health also pitching in resources and manpower to execute inoculation after it is flagged off by Prime Minister Narendra Modi on the morning of January 16 [See box: How Non-Health Ministries are Helping].
![vaccine_7 vaccine_7]()
The vaccine doses left the manufacturing facilities on January 12 for different parts of the country. Companies like Kool-ex, a logistics company from Pune, and airports and airlines assisted in reaching the doses to various state governments. “The government has defined the structure as of now,” Kunal Agarwal, director at Kool-ex, told Forbes India on the day of the vaccine transit. “There are going to be several government depots where the manufacturer has to send these vaccines. From there on, the government would be handling the movement of the secondary and last-mile sections.”
![vaccine_3 vaccine_3]()
On January 13, the Chhatrapati Shivaji Maharaj International Airport (CSMIA) saw the transportation of a total of 227 boxes, containing approximately 2,72,400 doses of the vaccine, to be delivered to over 20 destinations across the country, including Agartala, Cochin, Raipur, Jammu, Srinagar, Gorakhpur, Dehradun, Jabalpur, Trivandrum and Indore. A spokesperson for the Delhi International Airport Limited (DIAL), told Forbes India in an email that India’s busiest airport with an annual cargo handling capacity if 1.8 million metric tonnes is “working with other stakeholders in the supply chain, including pharma companies, airlines, forwarders and temperature-controlled transport service providers for efficient, reliable and uninterrupted services for end users”.
Vaccine doses reached government centres across the country by January 14 and 15. “Our vaccine storage facilities in Bengaluru and Belgaum have the capacity to store up to 50 lakh doses each. The first consignment released by the Centre reached Bengaluru on January 12, and it had 54 boxes containing 6.47 lakh doses of Covishield vaccine, apart from 64,750 vials, each vial having 10 doses,” Karnataka Health Minister Dr Sudhakar told Forbes India over a phone call.
![vaccine_2 vaccine_2]()
On January 13, the second consignment of 1.47 lakh doses arrived at the cold storage facility in Belgaum, taking the total number of vaccine doses to be administered in Karnataka to 7.94 lakh. Out of these, for immediate inoculation, around 15,730 doses are allocated for central government health care workers in Karnataka, 7.75 lakh doses for state health care workers and around 2,580 doses for Armed Forces Medical Services.
Apart from Covishield, Karnataka has received 20,000 doses of Covaxin manufactured by Hyderabad-based Bharat Biotech. “We are assured support by the central government for more doses and syringes for Karnataka,” says Sudhakar, who is targeting inoculation of seven lakh state health care and frontline workers within the current quarter and is unsure about whether or not the state will have to bear vaccination expenses for priority groups beyond the 3 crore people whose inoculation expenses the Centre has undertaken. Sudhakar expects the vaccination process to be glitch-free on the 16th.
“We have identified 29,000 vaccination sites that are spread across the state, which includes primary and community health care centres, taluka hospitals and private hospitals. The two dry runs conducted earlier this month threw up a few minor technical glitches in the use of the Co-WIN application for registering vaccination data—like people not getting OTPs on their phones or their details not getting registered with the nodal officer present on site—but those have been eliminated,” the minister adds.
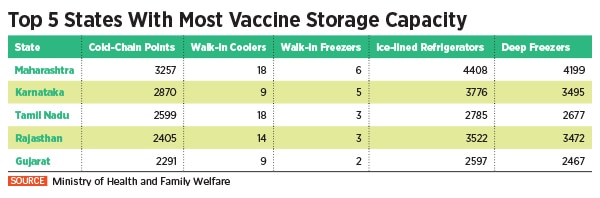
Other states are similarly prepared too. The Maharashtra government, which has the largest vaccination storage capacity in the country with approximately 3,257 cold chain points, has trained 18,000 vaccinators and intends to vaccinate over 3 crore people in three phases by August 2021. In Punjab, state health officials have said that details of as many as 1.6 lakh health workers targeted for first phase inoculation starting January 16 have already been registered on the Co-Win platform.
![vaccine_6 vaccine_6]()
In Tamil Nadu, health secretary J Radhakrishnan has said that the state has been allocated 5,56,500 doses of Covid-19 vaccines, which includes 5,36,500 doses of Covishield and 20,000 doses of Covaxin. On January 16, these doses will be administered across 307 sites, he added.
In Andhra Pradesh, vaccination will be officially launched at government hospitals in Vijayawada and Chinna Waltair, according to state government officials. The state has been allocated 4.99 lakh doses of the vaccine, including 4.77 lakh doses of Covishield and 20,000 doses of Covaxin, and doses will be administered at 332 session sites beginning January 16.
“The vaccines have been transported to all the cities and are ready for utilisation on the 16th. Whilst we can do all of these mock drills, only the reality of having mass lines and queues will test the entire system,” says Dr Kumud Kumar Dhital, surgical director for heart and lung transplantation and MCS Program at Yashoda Hospitals Hyderabad, which is the only private health care facility in Hyderabad that was selected to conduct Covid-19 vaccine dry runs or mock drills. The hospital will also be conducting vaccinations starting on January 16. “What’s important is will we be able to react to the situation and make things safe and workable, and that I think we can deliver,” he adds.
![vaccine_4 vaccine_4]()
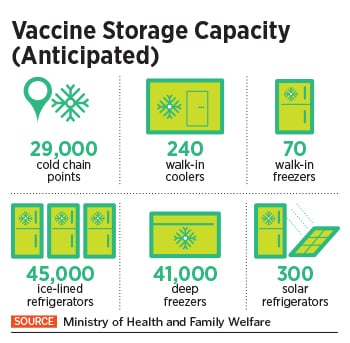
To undertake this Herculean task, the Indian government is piggybacking on its existing immunisation infrastructure including 29,000 cold chain points, 240 walk-in coolers, 70 walk-in freezers, 45,000 ice-lined refrigerators, 41,000 deep freezers and 300 solar refrigerators. The private sector, meanwhile, expects a bigger role as the government looks at the second phase of vaccination.
“The government is doing the last-mile delivery,” says Rampraveen Swaminathan, the CEO of Mahindra Logistics. “In the first phase, the government will control everything and we don’t see a large private sector participation. The private sector will come in in the second leg. The warehousing at the primary level isn’t a challenge, and so is the case with transportation. You will see capacity being ramped up in the next six months as things go ahead.”
Already, the central government has asked states and union territories to increase the number of vaccination session sites that would be operational every day, in a progressive manner. “States have been advised to organise vaccination sessions taking into account 10 percent reserve or wastage doses and average of 100 vaccinations per session per day,” the health ministry said. “Therefore, any undue haste on the parts of the states to organise unreasonable numbers of vaccination per site per day is not advised.”
From Hesitancy to Confidence
Meanwhile, even as the government looks at vaccination, especially since concerns have been raised about the efficacy of the vaccines, there might be some apprehension towards taking the vaccine.
![vaccine_5 vaccine_5]()
“Vaccination for Covid-19 is voluntary,” India’s health ministry had said earlier. “However, it is advisable to receive the complete schedule of the vaccine for protecting oneself against this disease and also to limit the spread to close contacts including family members, friends, relatives and co-workers.”
Dr Dhital, who is also a clinician and a researcher, has little doubt that India has the capability of vaccinating large populations in a quick and timely fashion. “The question is one of supply, and secondly, about uptick itself. There are still sections of the population that have issues about taking vaccinations.”
For instance, a dental surgeon in Navi Mumbai, who did not want to be identified, said that while he was registered in the first week of December to receive the vaccine, no information related to the actual vaccination procedure, or time and date of vaccination, has been provided yet. He also voices a concern around being administered Covaxin. “My primary concern currently is about the efficacy [of the vaccine]. I would have preferred a choice, especially knowing one of the two vaccines has not yet proven its efficacy in stage three clinical trials.”
Dhital agrees, saying that as a clinician and researcher, he feels that Bharat Biotech could have at least taken stock for a few more weeks. “At least until the entire enrollment of the 25,000 people or so that the company was aiming for clinical trials,” he says, adding that the public, therefore, has an understandable fear. “That said, Bharat Biotech has been consistently producing vaccines that have had no issues. And given Covaxin is being developed across a similar platform, along with ICMR and the National Institute of Virology, I am reassured.”
He, however, adds that the government needs to be more honest and straightforward with their communication. “I wish the heads of Bharat Biotech would not come out and say that their vaccine is 200 percent safe. That doesn’t help at all. You have to convince purists, and doctors, that everything is being done right so that we can convince our patients that vaccination is the wise thing to do and ask them to join the queue,” he explains. “We also need a little more honesty from the government. The population in India is largely well-versed to understand so they would rather have the truth than the government massaging the message.”
Karnataka health minister Sudhakar admits that he has also come across cases of vaccine hesitancy among people, including health and frontline workers. “Nobody can choose between the two vaccines, but the government has been working to communicate across levels and organisations that there will be no safety issues. Our DGCI [regulator] will never approve unless a vaccine is fully safe, secure and effective,” he says.
Nisha Chavan, police sub-inspector at the Kherwadi station in Mumbai, tells Forbes India that communication related to vaccinations from the police commissioner of the city came with an assurance that the vaccine is completely safe and “we should all take it”. Chavan, who worked through the lockdown to send migrant workers home and also contracted Covid-19 while on duty, informs that she and her colleagues have been registered to receive the vaccination and she is “happy about getting vaccinated”.
Mini, a nursing superintendent at the KJ Somaiya Hospital in Sion, Mumbai, received instructions on January 15 about how she will be vaccinated. “Although there is no clarity yet about if we need to go to the BMC [city municipal corporation] or they will come to our hospital, I don’t have any second thoughts about taking the vaccine shot. So far, we have only heard of [cases with] mild side-effects and I think it is important for us to take it.”
Clearly, the great Indian vaccination journey is only beginning.
(With inputs from Naandika Tripathi and Monica Bathija)