The race to make a Covid-19 vaccine in India
Companies in India are using diverse technologies to develop a coronavirus vaccine, but even as they race against time, the road to an efficacious solution is neither straight nor short


The timeline is historically short: Create in 18 to 24 months what you usually get five years or more to do. In mid-May, as the number of people testing positive for coronavirus increased to 4.48 million, including 82,000-odd from India, and counting worldwide, scientists have been breaking their backs to find a cure. A powerful treatment that experts say should be at least 95 percent effective against the outbreak and one that could be administered to people across ages, pre-existing health conditions, and geographies. Creating an effective coronavirus vaccine is a tall order, and an urgent one.
According to the Coalition for Epidemic Preparedness Innovations (CEPI), a foundation that tracks the global Covid-19 R&D landscape, there were at least 115 ongoing vaccine initiatives worldwide as of April 2020, out of which five are in early-stage clinical trials. In India, at least seven companies and research institutions are working on developing a vaccine candidate using different technological platforms and approaches. The CEPI notes that the “global vaccine R&D effort in response to the Covid-19 pandemic is unprecedented in terms of scale and speed”.
A Reuters report published on April 27 notes that historically, just 6 percent of all vaccine candidates end up making it to the market, often after a “years-long process that does not draw big investments until testing shows a product is likely to work”. But in the case of Covid-19, which has infected crores of people and killed lakhs across the world, while crippling the global economy, traditional processes are being sidelined, and governments, non-profits and pharmaceutical companies are investing billions into platforms even with low odds of success.
Rakesh Kumar Mishra, director of the Centre for Cellular and Molecular Biology (CCMB) in Hyderabad, says testing different approaches is key to achieving results in highly compressed timelines. At present, he explains, companies in India are using a wide range of methods (see Vaccines: Types and Methods) for vaccine development. This includes using the virus itself in a weakened or inactivated form, using nucleic acids (DNA and RNA) to prompt an immune response, or vaccines made using coronavirus proteins. Dr Krishna Ella and his team at Bharat Biotech in Hyderabad are working on ‘CoroFlu’, a one-drop nasal vaccine built on a pre-existing flu vaccine
Dr Krishna Ella and his team at Bharat Biotech in Hyderabad are working on ‘CoroFlu’, a one-drop nasal vaccine built on a pre-existing flu vaccine
Image: Dr Kaushik Ghosh For example, the three leading platforms being built in India, which have received funding from the department of biotechnology (DBT), use diverse approaches too. The Pune-based Serum Institute of India (SII), the world’s largest vaccine maker by volume, has undertaken mass production of the candidate being developed by the University of Oxford in the UK, which uses the weakened virus platform (see Indians in the Vaccine Race). Pharmaceutical major Zydus Cadila from Ahmedabad is using the virus vector approach, along with developing a DNA platform vaccine. Then there is Hyderabad-based Bharat Biotech, which is working on ‘CoroFlu’, a one-drop nasal vaccine built on a pre-existing flu vaccine.
“India has a huge opportunity because we have been a major vaccine exporter. Companies here have a robust capacity to manufacture vaccines at scale,” he tells Forbes India. The CCMB, which is one of the national laboratories of the government’s Council of Scientific and Industrial Research (CSIR), has been designated to validate antibody tests that companies develop for sale in India.Vaccine manufacturers say authorities have assured speeding up of regulatory processes. This means that if the feedback or permissions from regulators took 2-3 months earlier, it might be reduced to just a week or two now, giving companies significant headway.
“Basically, vaccine manufacturers want to go to Phase I of clinical trials as early as possible,” says Gautham Nadig, co-founder of pharma startup Mynvax, that is developing a protein-based vaccine in collaboration with the Indian Institute of Science (IISc) in Bengaluru, where it is incubated.
According to him, the real challenge will begin after animal trials, when, as part of clinical testing phases, the vaccine will have to be administered to humans. “Immunising a large group of volunteers to check whether the vaccine is effective will take time, but so long as safety is assured and you have efficacy data from animal testing, it gives a lot of hope.”
Scientists say while regulatory processes can be speeded up, biological aspects—like monitoring the immune response in a heterogenous population, fixing dosage and schedules of vaccine administration, and looking out for possibilities of other long-term adverse reactions to the vaccine—cannot be rushed.
“Without determining all this during clinical trials, going for large-scale vaccination will be risky,” says Dr E Sreekumar, a senior scientist at the Rajiv Gandhi Centre for Biotechnology (RGCB) in Kerala. He believes that once animal testing is successful, a realistic timeline for vaccine candidates to reach large-scale clinical use is at least a year. The safety and long-term efficacy of a vaccine is usually tested in a total of three such clinical phases (see How Safety and Efficacy are Determined).
However, given that time is of essence, it is up to the government and monitoring agencies to decide whether they want to be conservative or work out newer, faster, collective methods. “The government is actively working on streamlining and fast-tracking the process while adhering to safety norms. Because the quicker the vaccine is developed, the more beneficial it will be,” says Prabuddha Kundu, co-founder of Premas Biotech in Gurugram, which is developing a yeast-based protein Covid-19 vaccine.
 Mynvax Co-founders Raghavan Varadarajan (left) and Gautham Nadig are developing a vaccine in collaboration with the Indian Institute of Science, Bengaluru, where Mynvax is incubated
Mynvax Co-founders Raghavan Varadarajan (left) and Gautham Nadig are developing a vaccine in collaboration with the Indian Institute of Science, Bengaluru, where Mynvax is incubated
Image: Phani Rao
Path to the Vaccine
Many companies in India are building vaccine candidates in partnership with international organisations. The SII will jump-start manufacturing the vaccine developed by the University of Oxford at its own risk while human safety trials are underway whereas the company owned by billionaire Cyrus Poonawalla has partnered with US biotech firm Codagenix and Austrian firm Themis to work on two other platforms. The vaccine candidate being developed by Premas Biotech is licensed out to Akers Biosciences in the US, while the CoroFlu nasal vaccine by Bharat Biotech is in collaboration with virologists at the University of Wisconsin-Madison and vaccine maker FluGen in the US. Another Hyderabad-based company, Indian Immunologicals, is developing a vaccine candidate using a live attenuated virus by partnering with Griffith University in Australia.
Microsoft Founder Bill Gates, whose non-profit Bill & Melinda Gates Foundation is among the biggest funders of vaccines worldwide, estimates that at least 7 billion doses of coronavirus vaccine (double, in the case of a multi-dose vaccine) need to be manufactured and distributed to satisfy demand across the world. This, he suggests in a post on his blog GatesNotes, will require a “global cooperative effort like the world has never seen”.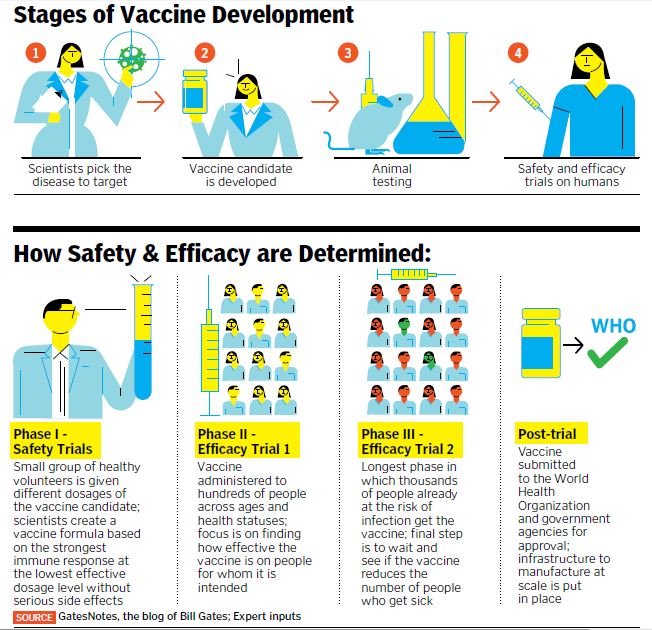 Vaccine manufacturers in India say international collaborations foster sharing of knowledge, infrastructure and expertise, and improve quality standards for faster results. Sreekumar of RGCB, however, points to a possible gap in vaccination coverage and immunisation, whereby the developed countries will ensure they use the bulk of the primary supplies while vaccines will be available in India only once their demand is satisfied. “So it would be important for us to have our own capacity too,” he says.
Vaccine manufacturers in India say international collaborations foster sharing of knowledge, infrastructure and expertise, and improve quality standards for faster results. Sreekumar of RGCB, however, points to a possible gap in vaccination coverage and immunisation, whereby the developed countries will ensure they use the bulk of the primary supplies while vaccines will be available in India only once their demand is satisfied. “So it would be important for us to have our own capacity too,” he says. Prabuddha Kundu (centre) and his team at Gurugram-based Premas Biotech are developing a yeast-based protein Covid-19 vaccine
Prabuddha Kundu (centre) and his team at Gurugram-based Premas Biotech are developing a yeast-based protein Covid-19 vaccine
Image: Nupur Mehrotra
On May 9, the Indian Council of Medical Research (ICMR) announced a collaboration with Bharat Biotech to develop a “fully indigenous vaccine for Covid-19”. An ICMR statement said the virus strain isolated at the National Institute of Virology in Pune has been transferred to Bharat Biotech’s labs to initiate vaccine development.
The Hyderabad-based company is also leading a project sanctioned by the CSIR to develop human monoclonal antibodies, which will block the spread of infection in the body by binding to the virus and making it ineffective. “We are fast-tracking the development process to make the antibodies available within the next six months, and thus improve the treatment efficacy,” says Dr Krishna Ella, chairman and managing director of Bharat Biotech. The company has successfully developed 10 different viral vaccines so far, including for H1N1 (swine flu), rabies, polio and the rotavirus. It has delivered more than 5 billion doses of viral vaccines. Risks and Bottlenecks
Risks and Bottlenecks
Vaccine manufacturers Forbes India spoke with say the lockdown posed immediate challenges in the development process, with disruptions in the supply chain and movement of people and scientists. Then there are also funding requirements to be met. For example, Mynvax has applied for funding from the Biotechnology Industry Research Assistance Council (BIRAC) and is awaiting a response. “The funding amount depends on how far we will take the vaccine forward on our own. For going all the way up to Phase I of clinical trials, we will require about ₹15 crore, but if we just do process development and let a manufacturer take up clinical trials and production, we would need just half the amount,” Nadig says.
The SII, according to media reports, will spend between ₹300 million and ₹400 million over the next five months to make around 3-5 million vaccine doses per month, apart from investing roughly ₹6 million on making a new manufacturing unit to solely produce coronavirus vaccines. It expects a price of ₹1,000 per vaccine.
Experts believe that being price-conscious will be important for India to make the vaccine more accessible, but the cost of the vaccine is likely to be influenced by a number of factors. “The price will be determined by factors, including the components used to create the vaccine, its easy adaptability and transfer to other countries, and finally, the method and quantity of dosage,” says Kundu of Premas Biotech.
Another major factor for delay could be the fact that while India is known to manufacture vaccines for international organisations and agencies, a lot of the indigenous research in this space had to be started from scratch, believes Sreekumar. “I don’t know how efficient we will be in transferring an indigenous vaccine candidate from lab to industry,” he says.
A vaccine should not be considered as a panacea, Sreekumar explains, since the coronavirus is fast-mutating and keeps changing. Also, there are possible issues of vaccination-induced adverse events such as antibody-dependent enhancement (ADE) of infection as documented in SARS vaccine studies. “From an evolutionary perspective, the new strains of the coronavirus are more diverse compared to the original strains in Wuhan, China. There are a lot of uncertainties,” he says. “Vaccine development will take time… meanwhile, we can have good anti-virals, revalidate and repurpose pre-approved drugs that have already been tested for safety profiles. The latter could be used immediately with supportive care,” he says.
The ICMR, for example, has identified drugs that could be repurposed, including remdesivir (originally developed to treat Ebola), Mycobacterium w (originally developed to treat leprosy) and lopinavir/ritonavir (a fixed-dose drug combination used for treatment and prevention of HIV/AIDS. Itolizumab, an anti-inflammatory psoriasis drug developed by pharma company Biocon, has also been permitted for use in moderate-to-severe Covid-19 cases, and is being trialled in Mumbai and Delhi. Senior scientist Dr E Sreekumar at the Rajiv Gandhi Centre for Biotechnology, which has been researching on the virus to aid vaccine developmentProduction of antivirals, which can reduce the life cycle of the virus and target its proteins, is also making headway in India, though safety and efficacy tests are yet to be done. On May 12, Noida-based Jubilant Life Sciences became the first company in India to sign a non-exclusive licensing agreement with US biopharma company Gilead Sciences to manufacture remdesivir, which, according to initial data, is a promising potential therapy for coronavirus. Glenmark Pharmaceuticals, meanwhile, has initiated clinical trials in India of its antiviral drug favipiravir.
Senior scientist Dr E Sreekumar at the Rajiv Gandhi Centre for Biotechnology, which has been researching on the virus to aid vaccine developmentProduction of antivirals, which can reduce the life cycle of the virus and target its proteins, is also making headway in India, though safety and efficacy tests are yet to be done. On May 12, Noida-based Jubilant Life Sciences became the first company in India to sign a non-exclusive licensing agreement with US biopharma company Gilead Sciences to manufacture remdesivir, which, according to initial data, is a promising potential therapy for coronavirus. Glenmark Pharmaceuticals, meanwhile, has initiated clinical trials in India of its antiviral drug favipiravir.
Biocon Chairman and Managing Director Kiran Mazumdar-Shaw has also been facilitating the development of blood plasma therapy [where a drug is created using the blood plasma containing antibodies from those who have recovered from a disease] for the treatment of Covid-19. While she did not respond to Forbes India’s queries on Biocon’s role in India’s vaccine development landscape, according to media reports, Mazumdar-Shaw has been asking state governments to start collecting blood from live convalescent blood donors to initiate plasma therapy.
Vaccine manufacturers in India agree that they have to be prepared for changes in their path at every turn. Experts say that while this process is underway, countries must continue strengthening the infrastructure for conducting more diagnostic and antibody testing. Suchitra Ella, co-founder of Bharat Biotech, tells Forbes India that every effort to build a vaccine is trial and error. “Be it repurposing old drugs, developing vaccines from scratch or blood therapies, everything has to go on simultaneously. There are no standards set in stone yet and we have to be prepared with whatever solutions we can come up with.”
First Published: May 21, 2020, 14:00
Subscribe Now