Behind a black curtain in a small room, a titanium sapphire laser is prepared to fire at a tiny and very surprising target: A half-centimetre glass window surgically implanted into the skull of a live mouse. If all goes right the laser will fire for a quadrillionth of a second while the mouse runs on a white, treadmill-like ball and watches a computer screen. Thanks to special dyes, certain brain cells will glow green if the mouse is using them, their image captured by cameras capable of detecting a single photon.
The point to all this Star Trek-style technology could not be more profound. That tiny tangle of tissue in the mouse’s skull turns nerve impulses from the rodent’s eyes into an interaction. Decoding that process would give scientists the first true window into how a mammalian brain experiences the world.
It has a secondary benefit, too. Looking over the contraption puts a big—and pretty rare—smile on the face of Paul Allen, the 59-year-old Microsoft co-founder who has plowed $500 million into the Allen Institute for Brain Science, a medical Manhattan Project that he hopes will dwarf his contribution as one of the founding fathers of software. The institute, scattered through three buildings in Seattle’s hip Fremont neighbourhood, is primarily focussed on creating tools, such as the mouse laser, which is technically a new type of microscope, that will allow scientists to understand how the soft, fleshy matter inside the human skull can give rise to the wondrous, mysterious creative power of the human mind.
“As an ex-programmer, I’m still just curious about how the brain functions, how that flow of information really happens,” says Allen in a rare interview, in a conference room overlooking an active ship canal.
“The thing you realise when you get into studying neuroscience, even a little bit, is that everything is connected to everything else. So it’s as if the brain is trying to use everything at its disposal—what it is seeing, what it is hearing, what is the temperature, past experience. It’s using all of this to try to compute what the animal should do next, whether that animal is a mouse or a human being.”
It’s heavy stuff, fuelled by curiosity and scientific ambition, made yet weightier by issues of mortality and the neuro fragility Allen’s own brain has been coping with. In June, his mother, Faye Allen, a schoolteacher who inculcated him with a love of books and knowledge, died of Alzheimer’s. “Any time you’ve seen a loved one…,” Allen says, trailing off. “You see their personality, everything that makes them human, slowly slipping away, and there is nothing you can do about it.” And Allen himself has waged a fight against stage four non-Hodgkin’s lymphoma, a deadly blood cancer that is now in remission. He is flush and energetic, juggling our interview with phone calls about one of his sports teams, and filled with urgency about his legacy.
His first $100 million investment in the Allen Institute resulted in a gigantic computer map of how genes work in the brains of mice, a tool that other scientists have used to pinpoint genes that may play a role in multiple sclerosis, memory and eating disorders in people. Another $100 million went to creating a similar map of the human brain, already resulting in new theories about how the brain works, as well as maps of the developing mouse brain and mouse spinal cord. These have become essential tools for neuroscientists everywhere.
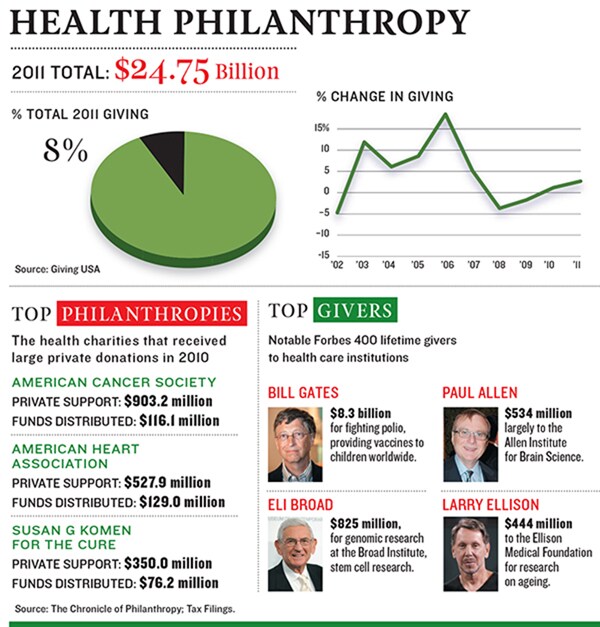
Now Allen, the 20th-richest man in America, with an estimated net worth of $15 billion, has committed another $300 million for projects that will make his institute more than just a maker of tools for other scientists, hiring several of the top minds in neuroscience to spearhead them. One effort will try to understand the mouse visual cortex as a way to understand how nerve cells work in brains in general. Other projects aim to isolate all the kinds of cells in the brain and use stem cells to learn how they develop. Scientists think there may be 1,000 of these basic building blocks, but they don’t even know that. “In software,” Allen says, “we call it reverse engineering.”
The willingness to fund these projects has gained Allen a growing number of disciples. “Paul has become a hero to me,” says David Anderson, a professor at Caltech who first proposed the mouse map project to Allen. “He’s done something for science in a way that very few other philanthropists have. It required that he have faith in science and go where his curiosity guides him.”
But there are also doubts about whether his new, grander plans will amount to anything. “The first phase of their investment really worked out,” says Susumu Tonegawa, an MIT professor and winner of a 1987 Nobel Prize who has done extensive brain research. But can Allen’s industrial approach really solve the mystery of how the brain creates consciousness? “It’s one of the biggest unresolved issues in brain research,” he says. “Whether it will work or not, I don’t know.”
Health issues have defined Allen’s career for the past three decades. He left Microsoft after beating Hodgkin’s lymphoma in 1982 and never returned. Bill Gates would turn Microsoft into one of the most essential companies of the 20th century, while Allen, whose shares continued to soar, spent billions on various passions. There were sports teams (the Portland Trail Blazers and the Seattle Seahawks), cable companies (Charter Communications, on which he lost $8 billion) and research labs (the for-profit Interval Research, shut down in 2000). There was the first privately-funded human spaceflight and the search for extraterrestrial life. There was the Gehry-designed EMP Museum, which the electric guitar buff originally built in part as a tribute to fellow Seattle native Jimi Hendrix. There was one of the largest yachts in the world, the Tatoosh, a 300-foot beauty with a 6-foot shaded swimming pool and a saloon that features a French limestone fireplace.
Allen began thinking about a big neuroscience project in the late 1990s, while he was making a flurry of investments in Seattle biotechnology companies. One, an outfit called Rosetta Inpharmatics, was doing genetic work that could be seen as a precursor of the institute’s Mouse Brain Atlas. Cancer researcher Stephen Friend, its chief executive, remembers long discussions about “a library of Alexandria for brain data”. He set up a meeting for Allen with James Watson, who won the Nobel Prize for co-discovering the structure of DNA.
Watson pushed Allen to think even bigger. Start an institute like Rockefeller University in New York or the Salk Institute in San Diego, he told him, and bring the best minds of science there. Allen balked, both at the potential $1 billion price tag and the memories of closing Interval Research, which failed partly because he had filled it with too many scientists merely completing their own projects.
But the sequencing of the human genome, an example of the power of industrial-scale science, inspired him, and he wanted to fund something similar. “I’m a person that did have the ability to do something that would jumpstart progress for scientists around the world,” he says.
But what? Allen decided to host a series of what he calls “charettes”, or brainstorming sessions, first in Seattle, and then on two separate cruises with scientists on the giant Tatoosh. Guests included Watson neuroscientist Richard Axel, another Nobel Laureate and Leland Hartwell, a third. All the scientists proposed ideas close to their own interests. The psychologist and linguist Steven Pinker argued for a ‘molecule to society’ institute that would study both neurology and behaviour. Others wanted to focus on experimental animals, like the C. elegans worm or primates.
But Caltech’s Anderson, a neuroscientist and geneticist, proposed the winning idea: A map of how genes work in the mouse brain. Every cell in a mouse—or, for that matter, a human—has the same 20,000 genes. What makes a heart cell different from a brain cell is how those genes are used.
And scientists can watch the genetic code being accessed like watching a computer reading a hard disk. In order to use a piece of genetic code, cells must transcribe it from DNA, which is stuck in the middle of the cell, to a messenger chemical called RNA. Scientists were learning how to measure RNA levels figuring out what parts of the mouse brain use which genes would help neuroscientists figure out what the genes do.
All of the scientists, Allen says, agreed that the project was worthwhile—despite every researcher having his own agenda. “It appealed to me because it was something that hadn’t been done, something that could be scaled, something that created a database that could be accessible worldwide and would lift all boats in the area of neuroscience,” he says. It also was similar to the role he’d played at Microsoft, where he had created emulators that allowed others to write software again, he was creating developer tools, this time for neuroscientists.
His mother, who loved books so much that when she was asked to name 100 favourites she could only narrow the list to 165, was suddenly veering from being able to do crossword puzzles one moment to being unable to remember what she had just said. In early 2003, she was diagnosed with Alzheimer’s. Allen wrote in his journal that he was “sick at heart”, and the Allen Institute for Brain Science was started in short order with the first $100 million donation.
![mg_67619_reverse_engineering_280x210.jpg mg_67619_reverse_engineering_280x210.jpg]()
Some philanthropists put their money towards buildings or infrastructure. Others channel it straight to the needy. Allen, in tackling the brain, spends it on talent. At the outset, he recruited Allan Jones, who had worked at the genetics startup, Rosetta, that Allen had dabbled with, to develop the Mouse Brain Atlas. Quickly, he took over the whole institute.
Instead of hiring big-name professors, Jones recruited a platoon of 60 young scientists taking breaks from graduate school or pit stops between academia and industry. Mouse brains were frozen and sliced by automated machines each slice fit on a single microscope slide. Technicians dipped each slice in an RNA solution that stained cells containing a single sequence of RNA in a particular colour. Only one gene can be captured per slice and six per brain, meaning it took 4,000 mice to create the Allen Brain Atlas, made available free on the web in December 2004 and largely finished in 2006. The completed atlas involved 85 million images on 250,000 slides, or 600 terabytes of data—half as big as the entire internet in 2003.
The Mouse Brain Atlas fast became a standard tool for neuroscientists in both industry and academia worldwide. In 2006, scientists outside Allen’s institute had used the atlas to find genes that might influence susceptibility to multiple sclerosis in humans, eating disorders and memory. Husseini K Manji, head of neuroscience research and development at Johnson & Johnson, says that his scientists scope out projects using the atlas’ exclusive gene data. For Ed Boyden, a neuroscientist at MIT, looking for genes allows him to figure out what experiments to conduct. One of the strains of lab mice developed by the project has even become one of the most popular in the US.
Outside the lab, this was a dark period for Allen. In 2008, a heart arrhythmia required valve replacement surgery. A month later, his lung filled with fluid, requiring another operation. When fluid built up in his other lung, doctors diagnosed late-stage non-Hodgkin’s lymphoma, which had spread beyond his lymph nodes. He dictated parts of his memoir, Idea Man, while suffering through the bone-deadening fatigue from chemotherapy. At the same time, Charter Communications, the cable company he backed, was going into bankruptcy.
His exploding neuroscience hub was a welcome reprieve. Allen loved peppering his scientists with questions and plotting a path for the future. With the success of the mouse atlas, he put another $100 million towards a tougher proposition, an atlas of the human brain, which is 3,000 times as big, with 1,000 times as many cells.
All the equipment for slicing and staining the brains had to be refitted. And, more critically, the brains themselves were hard to acquire. Rather than 1,000 human brains, the map would have to be built with fewer than 10, which needed to come from people who had died in the prime of life without the brain being damaged by injury or illness.
Four years later, six brains have been donated and four analysed to some degree. The project is due to be finished this year, but the first brain images, put on line in 2010, are already yielding scientific results. A paper in Nature shows that the gene expression from the first two human brains in the new atlas varies only a little, yielding hope that scientists will be able to understand some of what it all means.
How might this work? A young University of California, San Francisco neuroscientist named Bradley Voytek used software to match words that frequently appeared together in the scientific literature with matches of where genes are expressed in the Allen atlas. For instance, he found that scientists studying serotonin, the neurotransmitter hit by Prozac and Zoloft, were ignoring two brain areas where the chemical was expressed in their research. It might even play a role in migraines. This data-driven approach led to 800 new ideas about how the brain may work that scientists can now test, leading to hope that computational methods can help decipher the computer in our heads.
Understanding the brain, Allen argues, is much like being a medieval blacksmith trying to reverse engineer a jet plane. It’s not just that you don’t understand how the wing attaches to the fuselage or what makes the engine go. You don’t even know the basic theory of how air going over a wing creates lift. “Moore’s law-based technology is so much easier than neuroscience,” Allen says. “The brain works in such a different way from the way a computer does. The computer is a very regular structure. It’s very uniform. It’s got a bunch of memory, and it’s got a little element that computes bits of memory and combines them with each other and puts them back somewhere. It’s a very simple thing. So for someone to learn how to programme a computer, in most cases, a human being can do it. You can start programming. I did it in high school. Me and Bill Gates and our friends did that. Probably in a few months we were programming and probably understood what there was to understand about computing within a few years of diving into it.”
![mg_67623_allen_paul_280x210.jpg mg_67623_allen_paul_280x210.jpg]()
In the human brain, designed by evolution, every tiny part is very different from every other tiny part. “It’s hideously complex,” Allen says. And it’s going to take “decades and decades” of more research to understand. “We are talking about dozens and dozens of Nobel Prizes,” he says, “that have yet to be won to understand how the brain works.”
Despite the size of the challenge, Allen is undeterred. Shifting from his army of pit-stopping young scientists, in the last 18 months, he has hired three of the top neuroscientists in the country—and tripled the total head count at the institute to 200 with plans to double it yet again and move to a new, bigger building.
Christof Koch of Caltech, a former physicist with an Apple Computer logo tattooed on his left arm, says his colleagues think that he is crazy to leave the safety of academic tenure, but there’s no other way to get the job done. There are 10,000 neuroscience labs out there, but nobody else is trying to do anything as big. “We have 10-year plans to do something that can’t be done at the university,” he says.
Clay Reid, who quit a tenured job at Harvard, studies the mouse visual cortex. “I can have big dreams about what to do next, but managing those would be just impossible anywhere else.” This project, dubbed Mindscope, uses tools like the laser microscope that Allen saw for the first time when I met him to build a computer model of mouse vision. Colombian neuroscientist Ricardo Dolmetsch, on leave from Stanford, heads up another challenge: Using stem cells to figure out what the brain is made of. “If a mechanic didn’t know how many parts were in a car, would you trust that mechanic?” quips Koch, who likens current drugs for depression and schizophrenia to fixing that car by pouring oil over the top and hoping some gets into the motor.
In rolling up all this talent, Allen, the consummate entrepreneur and outsider, has switched sides. Science, like the early days of software, has generally been practised by hundreds or thousands of individual scientists working independently, competing to scoop one another—even large-scale research labs like Salk and Rockefeller work this way. Allen’s industrialised approach is known by researchers as ‘big science’, a worrisome trend to some. “The state of funding in neuroscience is worse than it’s ever been in my career,” says NYU neuroscientist Tony Movshon. “I think small, investigator-initiated science is the best way to get creative ideas.”
It’s possible that even the Human Genome Project that inspired Allen in the first place also slowed down the commercialisation of DNA sequencing technology, because it created a monopoly for machines made by one company, Applied Biosciences. Only after the project ended, and new, cheaper entrants entered the field, did we get a new revolution in genetics.
“There are going to be things where you need to have the standards and you need to do something systematically on a big scale,” shrugs Allan Jones, the Allen Institute’s chief executive. “Small labs don’t lend themselves to doing that well.”
In physics, he points out, it’s standard practice to get everyone on the same page and do large projects like CERN’s supercollider. There’s no reason that mysteries of the mind should require less of a group effort than mysteries of the universe.
Paul Allen is still more sanguine. “The proof will come a few years up the road, when we see the results of these new initiatives on which we are embarking now,” he says.
His $300 million investment was made with a five-year time horizon, but Allen and his team don’t talk in five-year or even 10-year time—they’re looking ahead decades. Allen, who says he feels great and is cancer-free, says he will contribute indefinitely as his scientists continue to deliver—and has even made plans to fund the institute after his death. “A big part of my own financial legacy,” he says, “is allocated to this kind of work for the future.”