Eyestem: A vision for the future
Medical entrepreneurs at the Bengaluru-based Eyestem are developing what they hope will be a platform that will bring cell therapy to the masses


There are some eye diseases that don’t have any cure today, and others where a single injection can cost ₹2 crore, says Jogin Desai, co-founder of Eyestem.
He and his co-founder Rajani Battu got talking about how to use the current tools available and create therapies that cost, say $10,000 a shot, instead. Thus, was born Eyestem. The idea is to make cell and gene therapy accessible to many more people.
The first product Eyestem is developing is to treat a condition called dry age-related macular degeneration, the largest cause of incurable blindness for people over 50 in the world. There are 170 million people in the world who suffer from it, with 40 million out of them in India.
The medical entrepreneurs at Eyestem have created “a suspension of cells" which could potentially reverse at least arrest vision loss and maybe even reverse vision loss as well, Desai says. One of the biggest milestones they’ve crossed over the last 12 months is that “we have now all the data that we need for a regulatory filing", which will allow them to progress towards human trials.
 “My hope is that once we get safety and efficacy out of this particular product, which will take about 12 to 15 months from the time we start, we should be able to get provisional commercial approval." Realistically that means the first commercial product from Eyestem could be ready by late 2025, he says.
“My hope is that once we get safety and efficacy out of this particular product, which will take about 12 to 15 months from the time we start, we should be able to get provisional commercial approval." Realistically that means the first commercial product from Eyestem could be ready by late 2025, he says.
In practice, the therapy is administered in the form of an injection and every hospital would be a potential customer if it has a qualified eye surgeon, who number about 700 in the country, when it comes to retina surgery, Desai says.
The aim of the treatment is to help rebuild a layer known as the retinal pigment epithelium, which is like the foundation on top of which our light-sensing cells are built.
“So, what we have done is created those cells in the lab and productised that science," Desai says. “From 1 million cells of starting material, we are able to create 48 million (cells) of our product."
An important part of the commercialisation journey will be to start a clinical trial in the US. There are two main types of age-related macular degeneration (AMD)—dry and wet. The latter is treatable through existing therapies, which are very expensive, and that market alone is about $9 billion, Desai says.
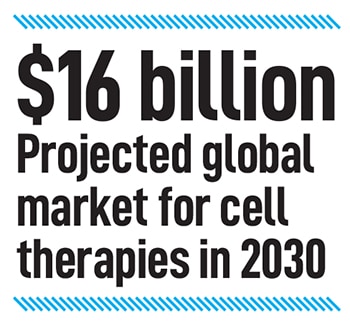 “Dry AMD has no cure anywhere in the world, whether you go to Boston or Bengaluru," he says. But it accounts for 85-90 percent of the cases of AMD, “so now you can see the potential addressable market and the business case," he says.
“Dry AMD has no cure anywhere in the world, whether you go to Boston or Bengaluru," he says. But it accounts for 85-90 percent of the cases of AMD, “so now you can see the potential addressable market and the business case," he says.
And the real success of Eyestem will come in five to 10 years, he says, because “what we are looking at is creating a cell therapy platform which is not just able to create one cell but is essentially able to create any cell of the body".
First Published: Oct 11, 2023, 13:46
Subscribe Now